Full HTML
Gastric Plexiform Fibromyxoma: A Narrative Review of a Rare Mesenchymal Tumor
Sajad Ahmad Salati1, Ajaz Ahmad Rather2
Author Affiliation
1 Professor, Department of Surgery, College of Medicine, Qassim University, Buraydah, Saudi Arabia
2 Professor, Department of Surgery, SKIMS Medical College, Srinagar, Jammu and Kashmir, India
Abstract
Gastric plexiform fibromyxoma (GPF) is a rare mesenchymal tumor that primarily affects the stomach, and to date, only about 170 cases have been reported. It is benign in nature with a favorable long-term prognosis. GPF mimics many disease entities, including the malignant ones, and the proper diagnosis differentiation requires a battery of investigations, including imaging, histopathology, immunochemistry, and molecular genetics studies. Management is generally surgical. This article is drafted to revisit various aspects of GPF to enhance the level of awareness in healthcare providers.
DOI: 10.63475/yjm.v4i3.0216
Keywords: Benign gastric tumor, gastric plexiform fibromyxoma, gastrointestinal stromal tumor, plexiform angiomyxoid myofibroblastic tumor, immunohistochemistry
Pages: 507-513
View: 14
Download: 18
DOI URL: https://doi.org/10.63475/yjm.v4i3.0216
Publish Date: 31-12-2025
Full Text
Gastric plexiform fibromyxoma (GPF) is a rare mesenchymal tumor that primarily affects the stomach. [1,2] The pathogenesis and molecular alterations of GPF are mostly unknown because of its rarity. [3] Following its initial description in 2007, about 170 case reports have been reported in peer-reviewed English-language literature. The condition is benign, but to establish a diagnosis, it must be distinguished from the many other malignant breast lesions that closely mimic it due to their molecular, histological, and radiological characteristics. [4–6] Given the extreme rarity of these tumors, awareness about this disease is limited, and hence, this article has been drafted to contribute to the existing literature and review the pathogenesis, clinical presentation, investigations, treatment, and outcomes.
This narrative review was conducted by searching peer-reviewed, English-language literature to consolidate existing knowledge on Gastric Plexiform Fibromyxoma (GPF). The search spanned from the initial identification of the tumor in 2007 through late 2025.
The review process involved:
- Case Identification: Reviewing the reported cases to determine epidemiological trends such as age, gender, and common tumor locations.
- Literature Analysis: Evaluating clinical data regarding various presentations, including incidental findings, dyspepsia, and acute complications like perforation.
- Diagnostic Review: Synthesizing findings from multiple diagnostic modalities, including Computed Tomography (CT), PET-CT, Magnetic Resonance Imaging (MRI), and endoscopic ultrasound.
- Pathological Assessment: Summarizing the established histopathological characteristics, immunohistochemical staining patterns, and recent molecular genetic discoveries.
- Management and Prognosis: Collating surgical approaches and follow-up data to define the standard of care and longterm biological behavior of the tumor.
Takahashi et al. first reported GPF in 2007 under the name “plexiform angiomyxoid myofibroblastic tumor” when they described submucosal gastric tumors in two patients: a 68-year-old man who had a 4.5 cm mass discovered by chance during laparoscopic cholecystectomy (LC) and a 50-year-old man who had a 4 cm mass that had perforated. [7] Histologically, the tumors displayed a plexiform growth pattern, extending from the serosa to the stomach wall’s submucosa. In 2008, Rau et al. published the third case of GPF. [8] Later in the same year, three more were reported with similar morphology and were referred to as “plexiform angiomyxoid tumor.” [9] In 2009, Miettinen et al. reported 12 additional cases and coined the term “plexiform fibromyxoma,” stating that it is a unique benign gastric antral neoplasm that should be distinguished from gastrointestinal stromal tumor (GIST), nerve sheath tumors, and other fibromyxoid neoplasms. [4] In 2010, the 4th edition of the World Health Organization’s classification of digestive system tumors approved the label “PF” and identified the tumor as a subtype of gastric mesenchymal tumors. [10] Since then, about 170 cases have been reported in peer-reviewed literature.
In most of the studies, GPF has been documented almost equally in men and women. Although most cases occur in the fifth decade of life, cases ranging in age from 5 to 81 years (median age, 46 years) have been reported. [11,12] The most common site for PF is the gastric antrum. [11–13] Tumor sizes range from 0.6 to 17.0 cm in the maximal diameter, with a median size of 4.0 cm. [14] Arslan et al. undertook a study to determine the frequency of PF in relation to GIST at Albany Medical Center, Albany, NY, and found that PF represents 1 to 2% (1.7%) of GIST-like tumors. They concluded that PF is an underestimated disease entity. [3]
The tumors are mostly found at the distal end of the stomach. In a review of literature conducted by Su et al., in 2019, out of the 114 GPF cases, 95 (83.3%), were located at the gastric antrum (including pylorus and gastric angle), followed, in decreasing order, by gastric body (N = 10; 8.7%), stomach (inside location unspecified, N = 5; 4.4%), gastric fundus (N = 4; 3.5%). [13]
Incidental Finding
In asymptomatic cases, PF may be incidentally found during upper gastrointestinal endoscopy, imaging, or an abdominal surgical operation. [15] In the initial case report that Takahashi et al. published in 2007, PF was detected incidentally in a 68-year-old man during LC. [7] Ebi et al. detected GPF in a 41-year-old asymptomatic woman when she underwent endoscopy during workup for thyroid-related disorder. [16]
Pei et al. reported a 45-year-old male patient in whom an abdominal lump was detected during an asymptomatic patient’s health examination, and abdominal computed tomography (CT) revealed a soft tissue mass (approximately 6 cm × 6.4 cm), which later on proved to be GPF. [17]
Dyspepsia
The more common symptoms include upper abdominal pain/discomfort, a feeling of fullness before finishing a meal, postprandial fullness, nausea, and vomiting. These symptoms are attributable to the distal gastric location.
Acute Abdomen
In the initial case report that Takahashi et al. published in 2007, a 50-year-old male had presented with an acute abdomen and, on exploration, was found to have a 4-cm GPF perforation. [7] Recently, in 2023, Mremi et al. also reported a 21-year-old female who presented with diffuse peritonitis secondary to GPF perforation. [18] Lee et al. reported GPF in a 42-year-old woman who presented with fever and acute abdominal pain. [19] CT scan revealed a fistulating abscess wherein there was a 12-cm cavitating pseudocyst-like mass in the gastric antrum, with fistulation to the gastric lumen.
Upper Gastrointestinal Bleeding
A patient of GPF may present with hematemesis/melena. [20] These symptoms may be attributable to the hypervascular nature of the lesions. Su et al. found that some amount of bleeding is present in about 41% of symptomatic patients, and on endoscopy in these patients, an ulcer is present on the tumor surface. [13]
Imaging for GPF is often challenging due to the nonspecific characteristics that resemble other gastric tumors, like GIST. [21] The key imaging characteristics include:
Computed Tomography (CT)
GPF may appear as a soft tissue mass with heterogeneous density. [21] Due to the increased vascularity of the tumor, contrast-enhanced computed tomography (CECT) often reveals mild enhancement of the solid portion during the arterial phase and strengthened progressive enhancement during the venous and delayed phases. [21] Munikrishnan et al. conducted a Positron emission tomography (PET)-CT in a 59-year-old female of GPF who had reported with a 6-month history of melena, early satiety, postprandial fullness, unintentional weight loss, and anorexia, and in Ultrasonography (USG) had shown an antral ulceroproliferative mass, and CECT had revealed an antropyloric exophytic mass. [22] PET-CT revealed a noticeably distended stomach, with a well-defined, non-homogeneously enhancing mass lesion involving the pyloric antral region (Figure 1).
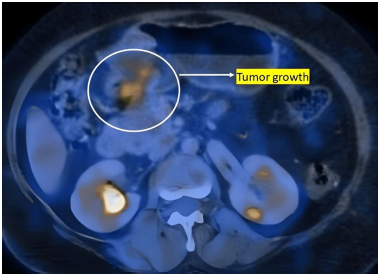
Figure 1: Axial PET-CT view demonstrating tumor growth. Image source: Munikrishnan et al. [22] Reused in accordance with the Creative Commons Attribution Non-Commercial (CC BY-NC 4.0) license.
Magnetic Resonance Imaging
GPF typically appears as a well-circumscribed mass. T1- weighted images often show low signal intensity, whereas the T2-weighted images show high signal intensity. Contrastenhanced images usually reveal heterogeneous and gradual enhancement, particularly in the delayed phase. [20]
Endoscopic Ultrasound
This modality can identify GPF as a submucosal mass with heterogeneous hypoechoic changes, originating from the submucosa or muscularis propria. [23]
GPF is detected as a submucosal mass by endoscopy, which is frequently the initial diagnostic procedure and occasionally results in endoscopic excision for cure. Although endoscopy reveals the size and location, the appearance may closely resemble other mesenchymal tumors. The usual findings include:
Visual Appearance
A well-defined or irregular, tan/pink, rubbery bulge, with a lobulated/nodular growth pattern and well-defined borders. [2,13,23]
Ulceration
The surface may be ulcerated, especially if it is large or has caused bleeding. [13,24]
Deformation
The tumor can cause deformation of the gastric lining. [23,24]
On gross examination, these lesions are not encapsulated and appear as multinodular, myxoid, or gelatinous masses (Figure 2) with or without hemorrhage, variably involving the intramucosal to subserosal and serosal parts of the stomach. [25] The tumor frequently exhibits tumor projection toward the serosal surface. [20,22,26]
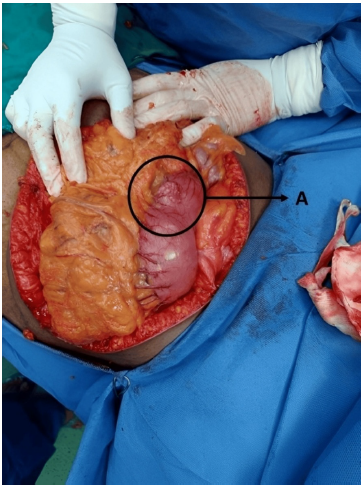
Figure 2: Intraoperative image showing nodular growth on the surface of the stomach. Label A indicates the nodular growth pattern. Image source: Munikrishnan et al. [22] Reused in accordance with the Creative Commons Attribution Non-Commercial (CC BY-NC 4.0) license.
At the microscopic level, GPF shows a multinodular, plexiform growth pattern with a proliferation of ovoid to spindle cells within myxoid stroma. Vascularity is increased, and spindle cells do not show any significant atypia. [13] Mitotic activity is rare (up to 7/50 High-power field (HPF)). Occasionally, more collagenous stroma is detected; this is a characteristic most frequently found in the extramural extension. The common features indicative of aggressive behavior, such as vascular and lymphatic invasion, are absent. Tumor necrosis is absent, though ulceration with surface necrosis has been reported in a few cases. [3,22]
The cells are positive for alpha-smooth muscle actin (α-SMA; Figure 3) and vimentin, and, partially, for CD10, h-caldesmon, and desmin. [27] The spindle cells are negative for epithelial membrane antigen (EMA), Discovered on gastrointestinal stromal tumors protein 1 (DOG-1), Protooncogene c-KIT (C-KIT), anaplastic lymphoma kinase (ALK; Figure 4), S-100, and hematopoietic progenitor cell antigen Cluster of differentiation 34 (CD34) with a low Kiel-67 (Ki-67) proliferation index. [20,28] The prominence of vascularity is highlighted by Cluster of differentiation 31 (CD31) and/or CD34. The myxoid stroma stains positively with Alcian Blue special stain. Focal keratin expression by the spindle cells has been documented in a few cases. [27]
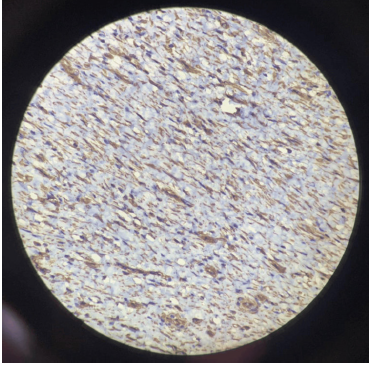 | 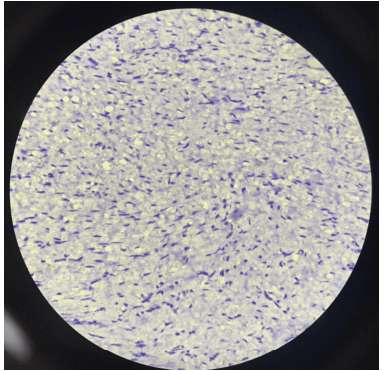 |
| Figure 3: Immunohistochemical staining shows positive expression for SMA. Image source: Munikrishnan et al. [22] Reused in accordance with the Creative Commons Attribution Non-Commercial (CC BY-NC 4.0) license. | Figure 4: Immunohistochemical staining shows negative expression for anaplastic lymphoma kinase (ALK). Image source: Munikrishnan et al. [22] Reused in accordance with the Creative Commons Attribution Non-Commercial (CC BYNC 4.0) license. |
GPF has not yet been linked to any particular molecular or genetic alterations. The C-KIT and Platelet-derived growth factor receptor A (PDGFRA) gene mutations, which define changes of GIST, are absent in GPF. [13] In a study conducted by Spans et al., 18% of cases of PF have been found to harbor Glioma-associated oncogene homolog 1 (GLI1) up-regulation and recurrent oncogenic fusion of MALAT1 (metastasis-associated lung adenocarcinoma transcript 1; in 11q12) with GLI1. [29] These alterations are similar to those displayed regularly in many other solid tumors, including gastroblastoma and malignant epithelioid tumors with GLI1 rearrangement. Banerjee S et al. also demonstrated that approximately one-third of GPFs have activation of the GLI1 oncogene, a transcription factor in the hedgehog (Hh) pathway, via a MALAT1-GLI1 fusion protein or GLI1 upregulation. [30] Eight cases of PFs with PTHC1 inactivation were described by Banerjee N et al. Bi-allelic chromosome 9q deletions of Protein patched homolog 1 (PTCH1) and Fanconi Anemia Complementation Group C (FANCC) were also observed in one case, and a partial PTCH1 deletion of exons 15– 24 on chromosome 9q in one case. Additionally, they documented a new link between PTCH1 inactivation and the onset of GPF. [31] Zhang et al. have recently reported co-amplification of GLI1, Cyclin-dependent kinase-4 (CDK4), and Mouse double minute 2 (MDM2) in GPF in a 36-year-old female. [2]
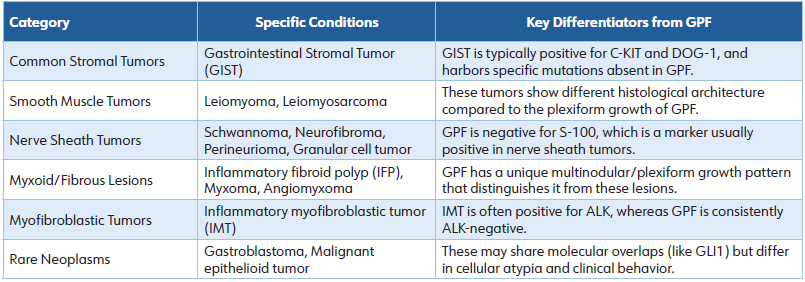
Differential Diagnoses
GPF mimics many mesenchymal disorders as depicted in Table 1 and is to be differentiated on the basis of histology, immunohistochemistry, and molecular genetics. [3]
Table 1: Differential Diagnoses of Gastric Plexiform Fibromyxoma
Surgical resection with negative resection margins is the treatment of choice. The size, location, and depth of the tumor typically dictate the resection approach, which is carried out using laparotomy, laparoscopy, and endoscopy. The procedures mentioned in literature include Partial gastrectomy, distal gastrectomy, subtotal gastrectomy, wedge resection, local resection, submucosal dissection, antrectomy, and total gastrectomy. [20] Only in a few cases is conservative management considered after endoscopic biopsy has been performed, and this option may be suitable for the elderly or selected patients with surgically contraindicated comorbidities. [20,31] Despite the benign pathological results, aggressive radical surgical treatment is recommended for the management of cases presenting serious clinical manifestations, such as perforation, infection, or significant hemorrhage, and with malignant suspicion, such as significant body weight loss or rapid tumor growth. [13]
The biological behavior of GPF is benign. Thus far, there have been no reports of local recurrence or distant metastases. [8,14] However, since the number of published cases is small, long-term observational studies with a substantial number of cases are still required to demonstrate definitively that GPF is benign. Most of the published cases of GPF had an uneventful postoperative phase, and among the cases that had followup periods, the uneventful or alive duration ranged from 0.75 to 396 months, with a median of 15 months and an average of 44.29 ± 72.5 months. [13]
Plexiform fibromyxoma is an extremely rare, benign mesenchymal tumor with differential diagnoses that include other gastric mesenchymal neoplasms. Overlap of histological and occasionally molecular characteristics can make diagnosis difficult in certain situations. The patients are generally asymptomatic and may be detected incidentally during evaluation of unrelated clinical situations or else present as dyspepsia, bleeding, or perforation. Surgical local excision is the main treatment, and the prognosis is good. LIST OF
ABBREVIATIONS
α-SMA alpha-smooth muscle actin
ALK anaplastic lymphoma kinase
CD31 cluster of differentiation 31
CD34 cluster of differentiation 34
CDK4 cyclin-dependent kinase-4
CECT contrast-enhanced computed tomography
C-KIT proto-oncogene
c-KIT DOG-1 discovered on gastrointestinal stromal tumors protein 1
CT computed tomography
EMA epithelial membrane antigen
FANCC Fanconi Anemia Complementation Group C
GIST gastrointestinal stromal tumor
GLI1 glioma-associated oncogene homolog 1
GPF gastric plexiform fibromyxoma
HPF high-power field
LC laparoscopic cholecystectomy
Ki-67 Kiel-67 MALAT1 metastasis-associated lung adenocarcinoma transcript 1
MDM2 mouse double minute 2
PDGFRA platelet-derived growth factor receptor A
PET positron emission tomography
PTCH1 protein patched homolog 1
USG ultrasonography
ACKNOWLEDGEMENTS
Figures 1 to 4 have been reused from Munikrishnan et al. [22] under the terms of the Creative Commons Attribution Non-Commercial 4.0 International License. https:// creativecommons.org/licenses/by/4.0/ and the source has been clearly cited in the labels of the figures. The author expresses gratitude to the publishers for having kept the article available for Open Access.
AUTHORS’ CONTRIBUTION
All authors have significantly contributed to the work, whether by conducting literature searches, drafting, revising, or critically reviewing the article. They have given their final approval of the version to be published, have agreed with the journal to which the article has been submitted, and agree to be accountable for all aspects of the work.
SOURCE OF FUNDING
None.
CONFLICT OF INTEREST
None.
References
1. Takahashi Y, Suzuki M, Fukusato T. Plexiform angiomyxoid myofibroblastic tumor of the stomach. World J Gastroenterol. 2010;16(23):2835-2840.
2. Zhang S, Yang Y, Li J, Li Z, Li W, Shi S. GLI1, CDK4, and MDM2 co-amplification gastric plexiform fibromyxoma: a case report and literature review. Genes Chromosomes Cancer. 2024;63(11):e70005.
3. Arslan ME, Li H, Fu Z, Jennings TA, Lee H. Plexiform fibromyxoma: Review of rare mesenchymal gastric neoplasm and its differential diagnosis. World J Gastrointest Oncol. 2021;13(5):409-423.
4. Miettinen M, Makhlouf HR, Sobin LH, Lasota J. Plexiform fibromyxoma: A distinctive benign gastric antral neoplasm not to be confused with a myxoid GIST. Am J Surg Pathol. 2009;33(11):1624-1632.
5. Lai J, Kresak JL, Cao D, Zhang D, Zhang S, Leon ME, et al. Gastric plexiform fibromyxoma: A great mimic of gastrointestinal stromal tumor (GIST) and diagnostic pitfalls. J Surg Res. 2019;239:76-82.
6. Li B, Zhang QF, Han YN, Ouyang L. Plexiform myxoid gastrointestinal stromal tumor: A potential diagnostic pitfall in pathological findings. Int J Clin Exp Pathol. 2015;8(10):13613-13618.
7. Takahashi Y, Shimizu S, Ishida T, Aita K, Toida S, Fukusato T, et al. Plexiform angiomyxoid myofibroblastic tumor of the stomach. Am J Surg Pathol. 2007;31(5):724-728.
8. Rau TT, Hartmann A, Dietmaier W, Schmitz J, Hohenberger W, Hofstaedter F, et al. Plexiform angiomyxoid myofibroblastic tumour: Differential diagnosis of gastrointestinal stromal tumour in the stomach. J Clin Pathol. 2008;61(10):1136-1137.
9. Galant C, Rousseau E, Ho Minh Duc DK, Pauwels P. Re: Plexiform angiomyxoid myofibroblastic tumor of the stomach. Am J Surg Pathol. 2008;32(12):1910; author reply 1912-1913.
10. Miettinen M, Fletcher CD, Kindblom LG, Tsui WM. Mesenchymal tumors of the stomach. In: Bosman FT, Carneiro F, Hruban RH, Theise ND, eds. WHO Classification of Tumours of the Digestive System. 4th ed. Lyon: IARC; 2010. p.74-79.
11. Morris MW, Sullivan L, Sawaya DE, Steiner MA, Nowicki MJ. Gastric plexiform fibromyxoma tumor in a child - Case report and review of the literature. J Pediatr Surg Case Rep. 2016;4:38-41.
12. Duckworth LV, Gonzalez RS, Martelli M, Liu C, Coffin CM, Reith JD. Plexiform fibromyxoma: Report of two pediatric cases and review of the literature. Pediatr Dev Pathol. 2014;17(1):21-27.
13. Su HA, Yen HH, Chen CJ. An update on clinicopathological and molecular features of plexiform fibromyxoma. Can J Gastroenterol Hepatol. 2019;2019:3960920.
14. Zhao X, Li X, Huang X, Shang L, Zhang J, Wu J. Gastric plexiform fibromyxoma resected by endoscopic submucosal dissection: A case report and review of literature. Hum Pathol Case Rep. 2021;23:200468.
15. Goyal S, Beso P, Khullar R, Sakhuja P. Plexiform fibromyxoma of the stomach: An underrecognized entity. Indian J Pathol Microbiol. 2023;66(2):343-346.
16. Ebi M, Nagao K, Sugiyama T, Yamamoto K, Saito T, Kurahashi S, et al. Gastric plexiform fibromyxoma resected using nonexposed endoscopic wall-inversion surgery: A case report. Case Rep Gastroenterol. 2022; 16(1):159-164.
17. Pei JY, Tan B, Liu P, Cao GH, Wang ZS, Qu LL. Gastric plexiform fibromyxoma: A case report. World J Clin Cases. 2020;8(22):5639-5644.
18. Mremi A, Nyoni V, Elisante J, Sadiq A, Nkoronko M. Viscus perforation as an initial presentation of plexiform fibromyxoma: A case report and review of the literature. Int J Surg Case Rep. 2023;108:107896.
19. Lee PW, Yau DT, Lau PP, Chan JK. Plexiform fibromyxoma (plexiform angiomyxoid myofibroblastic tumor) of stomach: An unusual presentation as a fistulating abscess. Int J Surg Pathol. 2014;22(3):286-290.
20. Szurian K, Till H, Amerstorfer E, Hinteregger N, Mischinger HJ, Liegl-Atzwanger B, et al. Rarity among benign gastric tumors: Plexiform fibromyxoma - report of two cases. World J Gastroenterol. 2017;23(31):5817-5822.
21. Yang MX, Zhao ZH, Yang JF, Chen B, Shen XZ, Wei JG, et al. Imaging findings of gastric plexiform fibromyxoma with a cystic change: A case report and review of literature. Medicine (Baltimore). 2017;96(52):e8967.
22. Munikrishnan DA, Mahalingam D, Anand R, Yudakar V, Viswanathan MV. A rare presentation of gastric plexiform fibromyxoma: Diagnostic challenges and surgical management. Cureus. 2025;17(7):e87318.
23. Kawara F, Tanaka S, Yamasaki T, Morita Y, Ohara Y, Okabe Y, et al. Gastric plexiform fibromyxoma resected by endoscopic submucosal dissection after observation of chronological changes: A case report. World J Gastrointest Oncol. 2017;9(6):263-267.
24. Nasralla A, Alwabari M, Alsaif O, Amr SS. Gastric plexiform fibromyxoma arising in the cardia in an adolescent male: A rare tumor with an unusual location. Case Rep Surg. 2020;2020:9037960.
25. Kim A, Bae YK, Shin HC, Choi JH. Plexiform angiomyxoid myofibroblastic tumor of the stomach: A case report. J Korean Med Sci. 2011;26(11):1508-1511.
26. Ikemura M, Maeda E, Hatao F, Aikou S, Seto Y, Fukayama M. Plexiform angiomyxoid myofibroblastic tumor (PAMT) of the stomach. A case report focusing on its characteristic growth pattern. Int J Clin Exp Pathol. 2014;7(2): 685-689.
27. Quero G, Musarra T, Carrato A, Fici M, Martini M, Dei Tos AP, et al. Unusual focal keratin expression in plexiform angiomyxoid myofibroblastic tumor: A case report and review of the literature. Medicine (Baltimore). 2016; 95(28):e4207.
28. Ma S, Wang J, Lu Z, Shi C, Yang D, Lin J. Plexiform fibromyxoma: A clinicopathological and immunohistochemical analysis of two cases with a literature review. J Int Med Res. 2021;49(8):3000605211027878.
29. Spans L, Fletcher CD, Antonescu CR, Rouquette A, Coindre JM, Sciot R, et al. Recurrent MALAT1-GLI1 oncogenic fusion and GLI1 up-regulation define a subset of plexiform fibromyxoma. J Pathol. 2016;239(3):335- 343.
30. Banerjee S, Corless CL, Miettinen MM, Noh S, Ustoy R, Davis JL, et al. Loss of the PTCH1 tumor suppressor defines a new subset of plexiform fibromyxoma. J Transl Med. 2019;17(1):246.
31. Banerjee N, Gupta S, Dash S, Ghosh S. Plexiform angiomyxoid myofibroblastic tumour of the duodenum: A rare entity. BMJ Case Rep. 2015;2015:bcr2015210004.
